[ad_1]
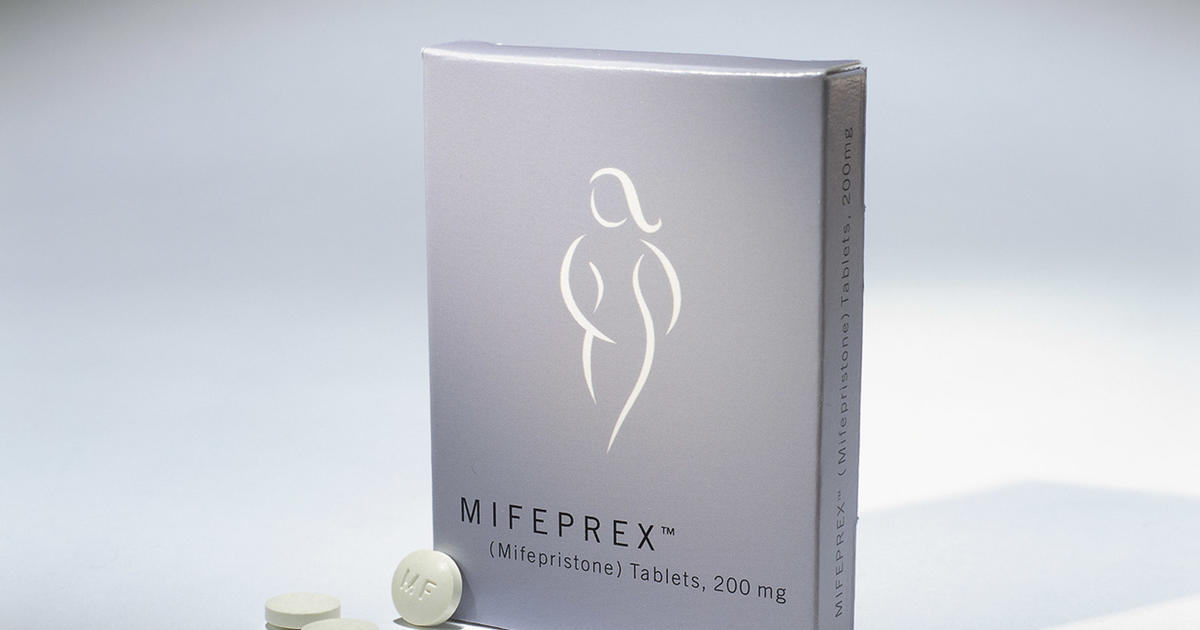
Washington — The Meals and Drug Administration on Tuesday finalized a rule change that broadens availability of abortion capsules to many extra pharmacies, together with giant chains and mail-order firms.
The Biden administration partially carried out the change final yr, asserting it will not implement a long-standing requirement that ladies decide up the drugs in individual. Tuesday’s motion formally updates the drug’s labeling to permit many extra retail pharmacies to dispense the capsules, as long as they full a certification course of.
The change might develop entry at each brick-and-mortar shops and on-line pharmacies. Ladies can get a prescription by way of telehealth session with a well being skilled, after which obtain the capsules via the mail, the place permitted by legislation.
Nonetheless, the rule change’s impression has been blunted by quite a few state legal guidelines limiting abortion broadly and the capsules particularly. Authorized consultants foresee years of courtroom battles over entry to the capsules, as abortion-rights proponents convey take a look at circumstances to problem state restrictions.
For greater than 20 years, the FDA labeling had restricted dishing out to a subset of specialty workplaces and clinics, as a consequence of security issues. Throughout the COVID-19 pandemic, the FDA quickly suspended the in-person requirement. The company later stated a brand new scientific assessment by company employees supported easing entry, concurring with quite a few medical societies that had lengthy stated the restriction wasn’t vital.
Two drugmakers that make brand-name and generic variations of abortion capsules requested the newest FDA label replace. Company guidelines require an organization to file an utility earlier than modifying dishing out restrictions on medication.
Danco Laboratories, which sells branded Mifeprex, stated in an announcement the change “is critically vital to increasing entry to treatment abortion companies and can present healthcare suppliers” with an alternative choice for prescribing the drug.
The American School of Obstetricians and Gynecologists known as the replace an “vital step” ahead.
“Though the FDA’s announcement at the moment is not going to clear up entry points for each individual looking for abortion care, it can permit extra sufferers who want mifepristone for treatment abortion further choices to safe this important drug,” the group stated in an announcement.
Greater than half of U.S. abortions at the moment are finished with capsules reasonably than surgical procedure, based on the Guttmacher Institute, a analysis group that helps abortion rights.
The FDA in 2000 permitted mifepristone to terminate pregnancies of as much as 10 weeks, when used with a second drug, misoprostol. Mifepristone is taken first to dilate the cervix and block the hormone progesterone, which is required to maintain a being pregnant. Misoprostol is taken 24 to 48 hours later, inflicting the uterus to contract and expel being pregnant tissue.
Bleeding is a standard facet impact, although severe issues are very uncommon. The FDA says greater than 3.7 million U.S. ladies have used mifepristone since its approval.
A number of FDA-mandated security necessities stay in impact, together with coaching necessities to certify that prescribers can present emergency care within the case of extreme bleeding. Pharmacies that dispense the capsules additionally want a certification.
[ad_2]
Source link










:max_bytes(150000):strip_icc()/Health-GettyImages-1477523726-d9489f5e044241b097588b0636bf7561.jpg)

















